
Depending on the phosphor composition, the effect can last from just a few seconds up to several hours. Persistent luminescence is usually observed only at room temperature (8) since, at higher temperatures, the stored energy is rapidly released, thus reducing the quality and efficiency of the phosphor. Furthermore, it is also interesting from a practical point of view and is commonly used in many different areas of applications such as emergency signaling, (1,2) biolabeling (3−5) or the creation of luminophores for white LEDs, (6,7) just to name a few. The phenomenon of persistent luminescence describes the release of energy stored by lattice defects located near the conduction band (CB). On the basis of spectroscopic measurements, the mechanism of persistent luminescence was constructed and discussed. The thermoluminescence measurements were done for the powders prepared using different methods to understand the impact of the synthesis conditions on the number and depths of the traps involved in persistent luminescence. The longest and brightest persistent luminescence was observed for the powders doped with 0.1 mol % (combustion) and 0.2 mol % of Ce 3+ ions (sol–gel). The most intense luminescence was observed for the powder with 0.5 mol % of Ce 3+ synthesized using the combustion method and 1 mol % in the case of the sol–gel sample. For all powders, persistent luminescence and emission decay processes were studied. Optical properties of the powders were examined using excitation, emission, and luminescence kinetic measurements. It was found that the synthesis method has a great impact on the morphology and, consequently, spectroscopic properties of the powders. The structure and morphology of the powders were characterized by X-ray diffraction (XRD) and scanning electron microscopy (SEM) techniques.
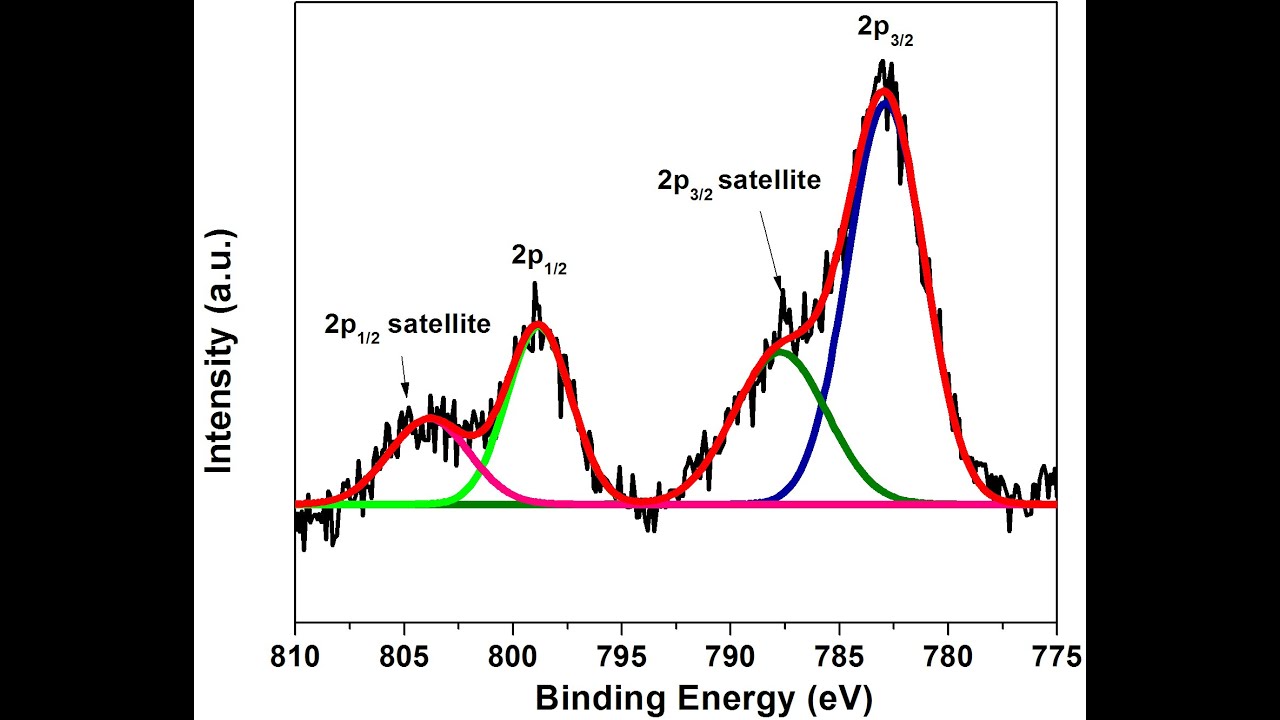
#Deconvolution of xps peak avantage software series#
The series of Gd 3– xCe xGa 3Al 2O 12 nanopowders doped with different concentrations of Ce 3+ ions were prepared by Pechini (sol–gel) and combustion methods.
